LiFePO4 6.4V 400mAh Battery for Medical Oxygen Sensors: Full Spec Guide for OEM Manufacturers
Author: Joan Li — Battery Engineer, Custom Pack Development | Himax Electronics Category: Technical Blog / Battery Engineering / Medical Applications
Introduction: When a Dead Battery Is Not an Option
Imagine a portable medical oxygen sensor going dark mid-shift in a busy ICU — or a wireless O2 monitoring node dropping offline during a home health visit. For medical device manufacturers, battery failure is not just an inconvenience. It is a patient safety risk, a warranty liability, and, in regulated markets, a compliance problem.
If you are currently specifying a power source for a medical oxygen sensor, a pulse oximeter, or a portable respiratory monitor, you already know that not every lithium battery belongs in a clinical setting. The chemistry, the protection circuit, the thermal behavior, the cycle life — all of it matters in ways that simply do not apply to consumer electronics.
This guide is written from the bench, not the marketing department. I am going to walk you through every parameter that affects your purchasing decision for the Himax LiFePO4 6.4V 400mAh battery pack (Model 110-00001) — and explain, in plain engineering terms, why each one matters for oxygen sensor medical applications.
Why LiFePO4 Is the Right Chemistry for Medical O2 Sensor Devices
Before diving into specifications, it is worth being direct about chemistry selection. OEM procurement teams frequently ask why we recommend LiFePO4 over standard NMC or LCO lithium-ion for medical o2 sensor products. The answer comes down to three factors that are non-negotiable in clinical and near-clinical environments.
Thermal stability. LiFePO4 cells have a significantly higher thermal runaway threshold than NMC chemistry. The phosphate-oxygen bond in the cathode is chemically stable, which means the cell does not release oxygen during breakdown the way NMC cells do. For a device strapped to a patient or carried in a clinical bag, this matters.
Flat discharge curve. LiFePO4 delivers a remarkably stable voltage output — around 3.2V per cell — for the vast majority of its discharge cycle before dropping off sharply at end-of-charge. For sensors that require consistent operating voltage to maintain measurement accuracy, this is a genuine engineering advantage over chemistries that slope continuously from full to empty.
Cycle life. Standard NMC batteries are commonly rated for 300–500 cycles before meaningful capacity loss. LiFePO4 routinely reaches 1,000–2,000+ cycles. For medical devices that are charged daily, that is the difference between replacing batteries once a year versus once every five or six years.
The 2S1P configuration of this pack — two 3.2V cells in series — gives you a nominal 6.4V output at 400mAh capacity, ideal for powering low-power medical sensor platforms where both stable voltage and compact form factor are required.

Complete Technical Specification Breakdown
This section is the specification data you need to complete a BOM entry, submit to your regulatory team, or pass to your mechanical engineer for integration planning. All values are sourced directly from Himax specification document HLFGB02 0A40-1527, Revision A2.
Cell-Level Specifications
The individual cells used in this pack are LiFePO4 format 14430, rated at 400mAh nominal capacity (minimum 370mAh) with a nominal cell voltage of 3.2V. Internal impedance is ≤60mΩ per cell, and cell dimensions are a maximum of 14.35mm × 43mm with an approximate weight of 14.5g per cell.
Battery Pack Electrical Parameters
| Parameter | Value |
| Pack Configuration | 2S1P |
| Nominal Voltage | 6.4V |
| Nominal Capacity | 400mAh |
| Minimum Capacity | 370mAh |
| Energy | 2.56Wh |
| Charge Voltage | 7.2V |
| Charge Method | CC/CV |
| Standard Charge Current | 0.08A |
| Max. Charge Current | 0.4A |
| Standard Discharge Current | 0.08A |
| Max. Continuous Discharge Current | 0.5A |
| Discharge Cut-off Voltage | 5.0V |
| Cycle Life | 2,000 cycles |
| Pack Internal Impedance | ≤350mΩ |
Physical Specifications
| Parameter | Value |
| Dimensions | Approx. 86.7 × 17.6 × 14.8mm |
| Weight | Approx. 32g |
| Output Wire | AWG28, (30+5)±3mm |
| Output Connector | Molex PicoBlade 1.25mm, 2-pin |
The Molex PicoBlade 1.25mm connector is a practical choice for medical device integration — it is a widely adopted, space-efficient connector that mates reliably with standard PCB footprints used across portable medical platforms.
Operating and Storage Temperature Ranges
| Condition | Temperature Range |
| Charging | 0°C to 45°C |
| Discharging | -20°C to 60°C |
| Storage | -10°C to 45°C |
For products deployed in ambulance environments, outdoor monitoring scenarios, or cold-storage adjacent settings, the -20°C discharge lower limit is a meaningful advantage over standard lithium-ion packs.
Electrical Performance Specifications
The electrical performance data below reflects test results under standard conditions: 20±5°C ambient temperature, 65±20% relative humidity.
Open-Circuit Voltage: ≥6.6V, measured within 24 hours of standard charge.
Battery Capacity Retention (Room Temperature): ≥95% of rated capacity after standard charge and 30-minute rest at 20±5°C.
Cycle Life Performance: ≥80% of initial capacity retained after 2,000 charge-discharge cycles at standard conditions. For a device charged once daily, this represents over five years of use before meaningful degradation.
Charge Retention (28-Day Storage): ≥95% of capacity retained after 28 days of storage at 20±5°C following standard charge. This supports products that may sit in warehouse inventory or hospital storage before deployment.
High-Temperature Performance (55°C): ≥90% capacity delivery after a 2-hour soak at 55°C, then discharged at standard rate. Relevant for devices used in warm clinical environments or transported in vehicles.
Low-Temperature Performance (-10°C): ≥50% capacity delivery after a 4-hour soak at -10°C. Cold-chain transport and outdoor monitoring scenarios should account for this in device power budgeting.
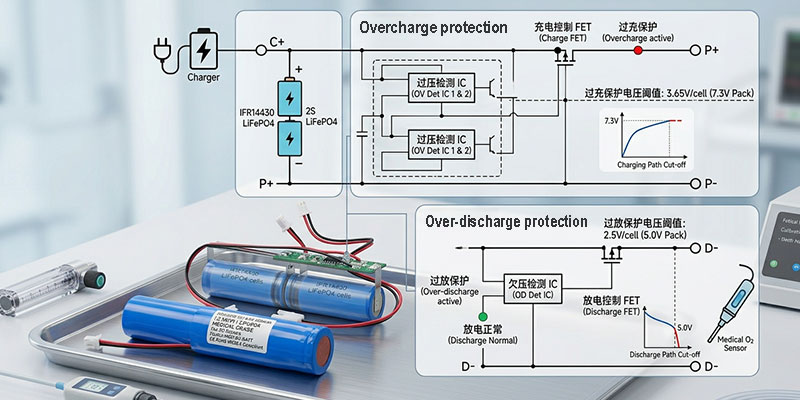
PCM Protection Circuit: The Safety Layer Your Compliance Team Will Ask About
Every battery sold into a medical-adjacent application should have a protection circuit module (PCM). The Himax pack integrates a PCM with the following parameters, all verified against the specification document:
Overcharge Protection
- Detect voltage: 3.65V ±0.025V per cell
- Delay time: 0.5–1.5 seconds
- Reset voltage: 3.45V ±0.05V per cell
Over-Discharge Protection
- Detect voltage: 2.0V ±0.08V per cell
- Delay time: 70–200ms
- Reset voltage: 2.5V ±0.1V per cell
Over-Current Protection
- Detect current: 2A ±0.5A
- Delay time: 5–40ms
- Reset: Release load
Short-Circuit Protection
- Condition: External short circuit detection
- Reset: Release load
PCM Resistance: ≤200mΩ
From an OEM perspective, the critical question is whether these protection parameters are compatible with your device’s charging circuit and BMS architecture. If your device uses a host-side charger operating at 7.2V with a current limit of 0.4A or less, this pack’s PCM is designed to work within that window. For applications with tighter current control requirements or non-standard charge profiles, contact our engineering team before committing to volume.
Safety and Mechanical Testing: What This Pack Has Been Through
Regulatory submissions for medical devices — whether CE marking in Europe or FDA clearance in the United States — require documented evidence that the battery does not create a hazard under foreseeable use and misuse conditions. This pack has been tested against GB/T18287-2013, UL1642, and CE61960 technology standards.
Crush Test: Force applied by a 32mm-diameter hydraulic piston to 13kN. Result: No fire, no explosion.
Drop Test: Dropped from 1 meter onto concrete in two orientations, twice each. Result: No explosion, no fire, no smoke.
Vibration Test: Simple harmonic motion at 1.6mm amplitude, swept from 10Hz to 55Hz at 1Hz per minute, applied for 30 minutes per axis across all three XYZ axes. Result: No leakage, no fire, no explosion.
Cell-Level Overcharge Test: Constant current at 1C to 4V per cell, then constant voltage hold until current reaches zero. Result: No explosion, no fire.
Cell-Level Short-Circuit Test: External short applied (≤50mΩ load) until voltage drops below 0.1V or cell surface temperature returns to ambient ±10°C. Result: No explosion, no fire, cell surface temperature below 150°C.
Heating Test: Cell heated in circulating air oven at 5±2°C/minute to 130°C, held for 30 minutes. Result: No explosion, no fire.
For procurement teams supporting FDA 510(k) submissions or CE technical files, these test results can be referenced in the battery section of your risk management documentation under ISO 14971.
Application Landscape: Where This Battery Fits in Medical Oxygen Sensing
The 6.4V 400mAh form factor was not chosen arbitrarily. It sits at the intersection of several design constraints that define portable oxygen sensor medical platforms: low continuous current draw, compact footprint, multi-day or multi-shift runtime, and the need to survive routine handling in clinical environments.
Here is where procurement teams most commonly deploy this pack:
Pulse Oximeters and SpO2 Monitors Handheld and wrist-mounted SpO2 monitors operate at low continuous power — typically well within the 0.5A maximum discharge current of this pack. The 2.56Wh energy capacity supports extended monitoring sessions, and the compact 86.7mm × 17.6mm × 14.8mm form factor integrates cleanly into handheld device enclosures.
Medical Oxygen Sensor Modules Dedicated oxygen sensor medical modules — including electrochemical O2 sensors used in anesthesia machines, ventilators, and gas monitors — require stable, clean power. The flat discharge profile of LiFePO4 reduces the need for additional voltage regulation circuitry in sensor analog front ends, which is a real BOM cost advantage.
Medical O2 Sensor Nodes in Wireless Monitoring Systems In hospital ward monitoring installations, distributed medical o2 sensor nodes that report to a central station need batteries that last through a full clinical shift without creating hot-swap logistical burdens. The 2,000-cycle rating means these nodes can be recharged nightly for years before battery replacement becomes a maintenance concern.
Portable Respiratory Function Analyzers Spirometers, peak flow meters, and portable capnography units in the 6V operating range benefit from the pack’s stable voltage and light weight. At approximately 32g, it does not meaningfully affect the ergonomics of handheld devices.
Home Health and Remote Patient Monitoring Devices Devices leaving the hospital environment face less controlled charging conditions. The robust over-discharge protection (2.0V detect with reset at 2.5V) prevents deep discharge damage in scenarios where patients or caregivers may not charge devices on a strict schedule.
Telemedicine Sensor Platforms and Wearable Vital Sign Monitors Low-power wireless vital sign sensors that integrate oxygen sensing, heart rate, and temperature monitoring in a single compact unit benefit from the pack’s combination of small size, adequate energy density, and safe chemistry.

OEM and Bulk Procurement: What to Verify Before You Commit to Volume
If you are sourcing this battery in quantity for production integration, here are the five technical verification checkpoints that matter most before purchase order placement.
- Connector CompatibilityVerify that your device’s PCB connector footprint matches the Molex PicoBlade 1.25mm 2-pin mating connector. If you require a different connector — JST PH 2.0, Hirose DF13, or a custom wire harness termination — that is a customization we can accommodate. Specify this at the RFQ stage.
- Charge Voltage CompatibilityYour device’s charging circuit must be configured to charge to 7.2V (±tolerance as specified by your charger IC). Charging to a higher voltage will trigger PCM overcharge cutoff on every cycle, reducing effective capacity and accelerating aging.
- Maximum Continuous Current MarginThe pack is rated for 0.5A maximum continuous discharge. If your device has peak current draws above this threshold — motors, pumps, high-power RF transmitters — validate your peak load against the PCM over-current trip point of 2A ±0.5A and ensure your duty cycle stays within the continuous rating.
- Temperature Range ValidationIf your product will be stored or operated outside the ranges listed in this specification, contact us before committing to this SKU. Operating the pack outside specified temperature limits will void the warranty and may affect regulatory compliance.
- Shipment Voltage and Pre-Integration StorageBatteries ship at 30–70% state of charge (SOV: 6.4–6.6V) as required for safe transport. If your assembly line will store batteries for more than three months between receipt and integration, plan for a top-up charge cycle before installation. Long-term storage at low state of charge accelerates capacity fade.
Delivery, Packaging, and Shipment Standards
Each pack is verified for voltage, internal resistance, and protective circuit function before shipment. Packs are transported at approximately 30–70% charge state in protective packaging designed to prevent mechanical stress during transit.
The packaging specification prohibits co-shipment with metal objects and requires protection from direct sunlight, moisture, severe vibration, and compression. Transport modes include road, rail, sea, and air freight under applicable dangerous goods regulations for lithium batteries (UN 3481).
If you receive a shipment and notice any abnormal odor — electrolyte smell in particular — do not use the affected units. Document and report the condition for warranty processing.
Customization Capabilities: When Standard Is Not Enough
The 110-00001 is a standard catalog pack. However, many medical device programs require something that does not exist off the shelf. Himax’s custom pack development capability covers:
- Connector type and orientation— including board-mount, right-angle, and custom wire harness terminations
- Wire length and gauge— tailored to your cable routing and connector placement within the enclosure
- Pack dimensions— within the constraints of the 2S1P 14430 cell configuration
- Capacity adjustment— alternative cell capacities in the 14430 form factor
- Label and marking— including custom branding, UL file references, and regulatory marking for your target markets
Custom development projects begin with an NDA and technical requirements document. Sample lead times for custom configurations are typically 3–5 weeks, with production tooling complete within 8–12 weeks depending on mechanical complexity.
Warranty and Quality Assurance
This battery pack carries a one-year warranty from the date of shipment. Himax will replace any unit where a defect is attributable to the manufacturing process. Warranty coverage does not extend to damage resulting from misuse, including charging outside specified parameters, mechanical abuse, immersion, or operation outside specified temperature ranges.
For quality-critical production programs, incoming inspection protocols should include voltage verification (target: 6.4–6.6V), impedance measurement (≤350mΩ at pack level), and a functional check of the protective circuit response to overcharge or over-discharge stimulus.
Safe Operation: A Note for Device Integration Teams
The following operating rules are non-negotiable from a safety and warranty standpoint. They are reproduced here for integration engineers who may be writing device-level operating instructions.
- Use only a LiFePO4-compatible charger rated for 7.2V / ≤0.4A. Do not use a generic lithium-ion charger calibrated for 8.4V (2S NMC).
- Do not continuously charge the pack for more than 8 hours.
- Do not reverse polarity. The connector is keyed to prevent this, but wire harness errors during custom integration are possible — verify polarity before applying power.
- Do not expose the pack to temperatures above 60°C during discharge or 45°C during charging.
- For storage longer than three months, maintain the pack at approximately 50% state of charge and store between -10°C and 45°C.
- Do not solder directly to the pack terminals or pierce the cells.
Summary: The Six Reasons Medical Device Manufacturers Choose This Pack
If you have read this far, you are likely evaluating this battery for a real program. Here is the short version for your decision brief:
- Chemistry match— LiFePO4 is the safest lithium chemistry available for medical-adjacent applications, with high thermal stability and no oxygen-releasing breakdown mechanism.
- Stable 6.4V platform— The 2S1P configuration delivers consistent voltage across 80%+ of the discharge curve, reducing downstream regulation requirements.
- 2,000-cycle rating— Five-plus years of daily charging without significant capacity loss, reducing lifetime cost and field service burden.
- Certified to GB/T18287-2013, UL1642, CE61960— The safety test documentation exists and is available to support your regulatory submission.
- Compact and light— 86.7mm × 17.6mm × 14.8mm, 32g, with a Molex PicoBlade connector for clean PCB integration.
- Customizable— If the standard pack does not fit your enclosure or connector requirements, we can build to your specification.
Talk to an Engineer Before You Place Your Order
Every medical device program is different. Before you commit to volume, I encourage you to send us your power requirements, connector specification, and operating environment parameters. We will validate that this pack — or a custom variant — is the right fit for your application, and we can provide sample units for your engineering validation testing.
- Website: himaxelectronics.com
- Tel: +86 (0)755-25629920
- Address: Building B, Nantong Avenue No.5, Tongle Community, Baolong Street, Longgang, Shenzhen, China
— Joan Li, Battery Engineer, Custom Pack Development, Himax Electronics
Specification data referenced in this article is sourced from Himax technical document HLFGB02 0A40-1527, Revision A2, dated September 20, 2024. All performance specifications are subject to the test conditions described in that document.


